There are little known cosmetic ingredients and then there are those that are more familiar. Parabens created a media explosion a decade ago that has resulted in nearly everyone at least knowing about these controversial compounds. While we don’t consider parabens acceptable for use in our formulations here at Blissoma, they certainly aren’t the only mainstream preservatives to avoid when shopping skincare.
Phenoxyethanol is a synthetic preservative that can be found in a wide range of skincare products. And while it's considered by some to be safer than parabens, this preservative has its good and bad points like any chemical. That’s why we’d like to shed light on the phenoxyethanol dangers that can exist, and the ways it interacts with your body.


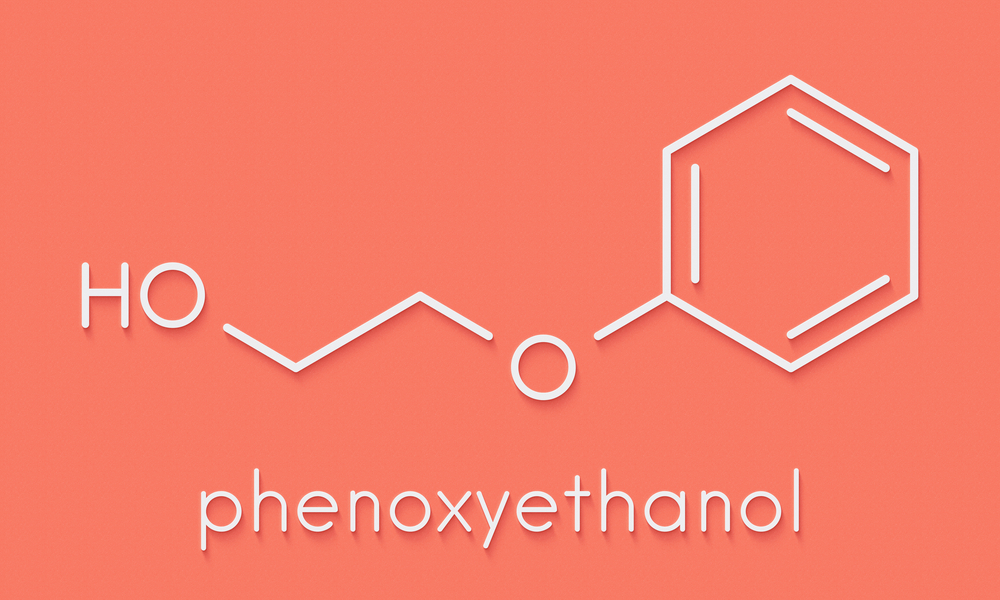
What Is Phenoxyethanol in Skincare?
Phenoxyethanol in cosmetics and personal care products is most commonly used as a synthetic preservative. This ingredient is produced for commercial use by treating phenol, a crystalline solid obtained from coal tar, with ethylene oxide, a carbolic acid. Both
coal tar and ethylene oxide are known to contain carcinogenic compounds but some feel the process of combining the two makes phenoxyethanol safe for use as a cosmetic preservative. It is from a class of chemical compounds called glycol ethers.
Phenoxyethanol occurs naturally in small amounts in green tea and chicory, but the version you will find in cosmetic products is always made in a lab. This makes it "nature identical" at best when found in your skincare. This issue creates interesting questions about what "natural" really is - it is a naturally occurring compound, is it a compound extracted directly from plants, or is it ok to synthesize it, and does the chemical feedstock matter? These issues are ones that the natural, green, and clean beauty world is still wrestling with.
We believe that the chemical feedstock absolutely matters, and that further the quantity of a compound that a person is likely to encounter in the natural world also matters. When a compound that occurs only in trace amounts in natural sources is highly concentrated in a lab and then used on human beings at levels they would not normally contact in nature it fundamentally changes how we are interacting with that substance. This issue has been explored previously with other preservative controversies like that around "Plantservative", the Japanese honeysuckle extract based preservative. Other compounds like undecane that are now sneaking into green beauty products have this same issue. The line for what is to be considered natural and what is non natural is currently a battleground and is likely to remain that way.
Phenoxyethanol is not allowed in cosmetic products applying for EcoCert or COSMOS certification. This already means that many natural-focused customers will choose to avoid this ingredient.
Not all cosmetic products require preservation. Oil blends are stable on their own but water-based products, like cleansers and lotions, need preservatives to protect against bacterial and microbial growth.
During the paraben-free craze it was common, and still is to a lesser degree, to see products labeled “paraben-free”. This was used by some mainstream beauty brands as a marketing effort to make their products seem more natural, less toxic, or safer.
The truth is, if a product needs to be preserved it cannot simply have preservative ingredients removed and be stable. If the parabens are removed from a skincare product, it would either need to contain other preservatives or be reformulated. And if a mainstream beauty brand reformulated its water-based products using natural preservation methods, they would most likely put that on the label rather than a mere “paraben-free”.
Paraben fear has prompted many cosmetics makers to use phenoxyethanol as a preservative. And while it may not have the reputation of parabens or carry the same potential risks, there are reasons to consider steering clear of this ingredient.
The best way to avoid synthetic preservatives is to read ingredient listings. Here’s how phenoxyethanol shows up on product labels: phenoxyethanol, 2-hydroxyethyl phenyl ether, 2-phenoxy- ethanol, 2-phenoxyethanol, 2-phenoxyethyl alcohol, ethanol, 2-phenoxy-, ethanol, 2phenoxy, ethylene glycol monophenyl ether, phenoxyethanol, and phenoxytol.
Contaminated skincare is a bad thing but synthetic preservatives have drawbacks too.
And skipping synthetics doesn’t mean you have to forgo safe products. There are many natural preservatives that can be utilized in their place for lotions, creams, and serums that have water content.
Green chemistry has continued to evolve in leaps and bounds and there are more choices of naturally based preservatives on the market for formulators than ever before. However the way that naturally based preservatives work is more complicated than many synthetic anti microbial ingredients, and they often cost more as raw materials. Usually a combination of several is needed in order to satisfy the needs of protecting a product from fungi and bacteria alike, which are sensitive to different things. More rigorous testing is needed to devise naturally based preservative systems that work in each individual product, which can also drive up development and laboratory costs. This can leave cosmetic manufacturers eager for the simplicity of using a broadly effective, comparatively "easy" ingredient like phenoxyethanol.
The preservative system we use at Blissoma varies by product. Oil based products don’t readily grow bacteria and fungi, and are kept fresh with antioxidants alone. We utilize a vegan preservation system for our facial care range, compiled from a customized combination of select ingredients after thorough testing. You can read more about the biodegradable, skin-friendly, safe and natural preservatives we use on our Ingredient FAQ page.
Is Phenoxyethanol Safe in Skincare?
A quick Google search on phenoxyethanol safety will likely leave you stumped. There are two camps when it comes to this issue – those who feel the ingredient poses no risks when used as directed in concentrations of 1% or lower and those who feel it’s best avoided.
You may want to note that Japan and the EU are the places where phenoxyethanol is currently restricted by law to be used at 1% or less. In the USA no such restrictions exist, nor is there any third party verification of ingredients content. This could mean that product manufacturers in the USA that use this ingredient may be using in excess of the advised 1% level, particularly if there are ingredients in a formulation that were preserved with phenoxyethanol prior to being added to the final recipe. If phenoxyethanol is then added to the final recipe at 1% this would put the total amount in the product over the 1% limit. It is nearly impossible to know if manufacturers are doing their math carefully to avoid this situation with no legal restrictions on them in the USA to cause them to act more cautiously.

As always, it’s a good idea to do your research when concerned about a cosmetic ingredient and to follow brands whose ingredient commitment you trust. Brands dedicated to creating synthetic-free products will avoid phenoxyethanol. We did some digging and found that this preservative comes with some potential unwanted side effects.
The Phenoxyethanol Dangers You Need to Know About
Properly preserved skincare is a priority for safety, but which preservatives we use and how often are important choices. Whether phenoxyethanol is safe or not is likely to be a personal decision for most people, and there is definitely some study data you will want to know about as you consider how you feel about this preservative.
A lot of the scientific study data available on phenoxyethanol is relatively old, and generally in the scientific world anything older than 10 years is very old since new testing methods and ideas are constantly evolving and new data is always being produced. With that in mind there are a few points of data you will want to know about.
Will phenoxyethanol cause skin reactions, contact allergies, and eczema?
Phenoxyethanol has been in use since the early 1980s, and very few incidents of contact allergy were reported in its early days of use. Reports of contact allergy increased in the 1990s and 2000s which could be simply due to its increased use in products. It could also represent growing sensitization in the population using it. However a 2011 study showed that phenoxyethanol showed one of the lowest risks of sensitization among preservatives evaluated in that study. The test group also included benzyl alcohol, parabens, and methylisothiazolinone (now known to cause contact allergy in many people). The sensitization exposure quotient (SEQ) was calculated by dividing the relative frequency of sensitization and the relative frequency of use. By this method phenoxyethanol was shown to have an SEQ of just .06 while methylisothiazolinone has an SEQ of 1.7. Some preservatives tested had SEQ values up to 9.0.
Some of the worst cases of skin sensitization that have been seen with phenoxyethanol occurred when it was being used as a component ingredient in the branded preservative blend known as Euxyl-K 400. The other chemical compound in Euxyl-K 400 is 1,2-dibromo-2.4-dicyanobutane. The sensitizing capability of this blend has been known since the early 1990s and most cosmetic manufacturers are probably not using this blended ingredient anymore.
All that said the SEQ calculated above was derived using data from a body of information collected between 2006-2009 and includes frequency of use as one of the criteria. The use of phenoxyethanol in cosmetics has absolutely risen over the last 10 years, meaning that those numbers could shift given that frequency of use would be way up. Frequency of contact contributes to sensitization. A 2017 review of the products listed in the American Contact Dermatitis Society Database showed that phenoxyethanol was found in 23.9% of common cosmetic products in the USA.
As well the data would have represented a broad section of people, which is great if you have "normal" skin. However if you are someone suffering from eczema or frequent contact allergies it could be far more likely that you could react to a substance like phenoxyethanol. Data collected from a typical population does not reflect reaction rates among exclusively people with highly reactive skin. So if you have highly reactive skin you may still wish to avoid phenoxyethanol.
Here are some of the cases where phenoxyethanol has caused skin reactions and contact dermatitis:
- A new 2021 paper describes a situation where a case of contact dermatitis on the hands of a man was attributed to phenoxyethanol exposure from hand cream.
- In 2015 a case was reported of skin reaction to an ultrasound gel with phenoxyethanol
- In 2009 a case of contact dermatitis occurred after exposure to an aftershave with phenoxyethanol.
- In 2008 a 42 year old woman had been experiencing hives anywhere she applied common body products, and had one episode of anaphylaxis. She was diagnosed as having a skin reaction to phenoxyethanol.
- In 2006 a positive scratch test for skin reaction to phenoxyethanol was reported after an 11 year old girl reacted to her mother's face moisturizer.
What happens when phenoxyethanol absorbs into your skin?
When phenoxyethanol is absorbed into skin it is metabolized in the liver to become phenoxyacetic acid and its conjugates. More than 90% of the metabolites are eliminated through urine. Very small amounts of phenoxyethanol may remain behind in organs and tissues, but this effect is thought to be minimal. Higher amounts of phenoxyethanol have been documented in women versus men and cosmetic products are thought to be the primary reason for this difference due to the increased number of personal care products used by women. About 47% to 85% of the phenoxyethanol applied to skin can absorb through skin and into the bloodstream. Because of this and the safety data of its damaging effects on the nervous system of infants women who are pregnant or breastfeeding should avoid phenoxyethanol. Phenoxyethanol is a nervous system depressant and could cause vomiting and diarrhea in infants, and the FDA has warned mothers not to use nipple cream containing phenoxyethanol.
The good news is that according to current study data available phenoxyethanol has been found to be at least non genotoxic (non mutagenic) and non carcinogenic. In one study using mice phenoxyethanol was found to be a mild reproductive toxin at high doses, and caused a decrease in litter size and weight.
How phenoxyethanol affects skin
While we thoroughly advocate for proper cosmetic preservation it cannot be understated that applying excess synthetic preservatives to the skin on a daily basis is shortsighted for the overall health of skin due to the way synthetic preservatives affect the skin microbiome.
A disrupted skin microbiome can lead to a wide variety of problems including dryness, eczema flares, breakouts, and more. Studies have found that cosmetic products and common synthetic preservatives are capable of having significant effects on the skin microbiome, including shifting the populations towards uncommon bacteria types and affecting overall diversity and abundance.
Skin that has an abundant, diverse population of microbes is healthier skin. One of the key reasons to use naturally based skincare and cosmetics is that truly natural products can have a much more beneficial effect on the skin microbiome. Adding synthetic preservatives to otherwise natural products compromises this inherent performance advantage.
Synthetic preservatives are valued in cosmetics because they work so well, and that performance remains true when a cosmetic product hits your skin. By contrast many natural preservatives have narrow performance requirements including specific Ph ranges they must be used at to be effective, specific concentrations they must reach, and other factors. This means that their preservative capabilities often break down significantly when cosmetics are applied to skin. This is more advantageous for the microbiome.
Many natural skincare products that are made without water also offer options to avoid synthetic preservatives. These are excellent choices for people looking to avoid additional challenges to their skin sensitivity, microbiome health, and overall body burden.
In short, preservatives play a hugely important role in the safety of cosmetics. But synthetic preservatives come with disadvantages to skin, the skin microbiome, and your overall wellbeing. We advise avoiding phenoxyethanol dangers by choosing plant-based skincare with natural preservatives.
Disclaimer: The information contained on this site is general in nature and for informational purposes. It is not meant to substitute for the advice provided by your own physician or other medical professional. None of the statements on this site are a recommendation as to how to treat any particular disease or health-related condition. If you suspect you have a disease or health-related condition of any kind, you should contact your health care professional immediately. Please read all product packaging carefully and consult with a healthcare professional before starting any diet, exercise, supplementation or medication program. Cosmetic products have not been evaluated by the Food and Drug Administration and are not intended to diagnose, treat, cure, or prevent disease.


